Pharmaceutical Sector Transformation: $4B Deals & $755M Investment
Author:
Intellectual Market Insights Research
Published Date:
11 Mar 2026
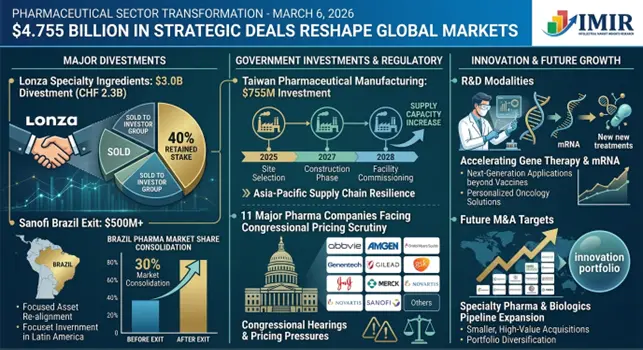
Pharmaceutical Sector Transformation: $4 Billion in Strategic Deals and $755 Million Government Investment Reshape Global Markets
On March 6, 2026, the pharmaceutical industry has made major strategic repositioning moves in which transactions of over 4 billion dollars were announced with huge government investments in its supply chains marking a historical change in manufacturing priorities, access to its markets, and regulatory measures. These processes indicate how industry leaders are gradually leaving commodity operations and governments are using massive capital to back pharmaceutical supply chains and increase the intensity of pricing transparency obligations.
- Lonza Executes $3 Billion Divestment to Accelerate High-Margin CDMO Strategy
In a deal worth CHF 2.3 billion (about $3 billion) the largest pharmaceutical portfolio restructuring deal of 2026, Swiss contract manufacturer Lonza announced the sale of its Capsules and Health Ingredients business to Lone Star Funds. The company will get CHF 1.7 billion cash payment at once and still own 40% stake with a potential outlay of CHF 3 billion at a later exit valuation of the company.
This deal radically changes Lonza into a pure-play contract development and manufacturing company in which it is only involved in high-margin biologics and advanced therapy manufacturing. The CHF 500 million share buyback scheme illustrates an immediate confidence by the management that there will be continued demand of complex manufacturing services where capacity bottlenecks will still be forcing the high premium prices on specialized manufacturing facilities.
Supply Chain Impact: The divestment causes direct tightening of the high-tech biologics manufacturing capacity because Lonza has all capital allocated to the expansion of complex molecule production. The drug developers who are in need of the production of antibody-drug conjugate, cell therapy, or gene therapy must anticipate the persistent capacity limitations and high premium pricing as the specialized CDMO players converge around the most valuable services. To prevent bottlenecks potentially causing delays in commercial launches, companies designing next-generation biologics should establish manufacturing alliances during earlier stages of development to circumvent any bottleneck.
Market Repositioning: As a business enterprise under Lone Star Funds ownership, Capsules and Health Ingredients business that has been split will more likely seek to achieve an operational efficiency enhancement and even consolidation with other consumer health manufacturing properties. This provides openings to companies that are interested in outsourcing dietary supplements and over-the-counter products to contract manufacturers, and may also heighten pricing pressure on the businesses that provide commodity pharmaceutical manufacturing services.
- Sanofi's $500 Million+ Brazilian Exit Creates 30% Market Share Consolidation
French drug manufacturer, Sanofi, sold its Brazilian generics business, Medley, to a local generics firm, EMS Pharma, at a price of in excess of 500 million dollars and formed a powerful generics player with an estimated 30 percent market share in the fast-growing generics market of Brazil. The underlying impact of such consolidation is the shape of competition in the largest pharmaceutical market in Latin America, where the adoption of generics is still growing rapidly because government healthcare policies favor the use of cheap medicine.
Market Concentration Effects: The EMS-Medley joint venture will possess vast sources of distribution channels, production capacity and product lines that will impose huge entry barriers to other smaller players. Such market concentration gives it unprecedented bargaining power with the public health care system in Brazil and the local health insurance companies, which may influence the pricing environment in the entire regional generics industry. Brazilian generic competitors with international generics manufacturers are willing to access their markets will better encounter a more consolidated competition that will be less responsive to significant market share growth.
Implications of Strategic Portfolio: Sanofi has been divesting volume-oriented business systematically to focus capital on innovative medicines in immunology, vaccines and rare diseases, which will allow the company to exit Brazilian generics. This firm has diverted its generics sales of more than 2 billion dollars to its high-research therapeutic markets where it enjoys patent and clinical differentiation that enables it to charge a higher price. This rationalization of portfolio approach proves how MNC pharmaceutical firms are putting sustainable competitive advantages ahead of revenue magnitude in commodity segments.
- Taiwan Commits $755 Million to Domestic Pharmaceutical Manufacturing Independence
The government of Taiwan declared a multi-year plan of 755 million to increase capacity in domestic pharmaceutical manufacturing, lessen its dependence on imported active pharmaceutical substances and create strategic stocks of medicines to react in times of emergency. This massive investment is a sign of increased international awareness that the pharmaceutical supply chain security has become a matter of vital national security concern in the wake of the COVID-19 disturbances that had revealed perilous reliance on concentrated manufacturing sectors.
Localization Acceleration of the Supply Chain: The initiative will be used directly to finance the development of new manufacturing plants, technology transfers to local firms as well as the establishment of local capacity to produce basic medicines that are currently imported into the country via China and India. Taiwan is not an exception, as it sends billions of dollars to the pharmaceutical supply chain localization similar to the United States, European Union, Japan, and other powerful economies, essentially disrupting the decades-old optimization of the industry to low-cost and centralized manufacturing centers.
Partnership and Investment Opportunities: Multinational pharmaceuticals are confronted with opportunities, and on the spot, to form either a technology transfer, joint venture, or local manufacturing business, with the government subsidizing, and with the business enjoying preference in the market. The firms that are ready to share the manufacturing knowledge and build up regional production capacity will benefit in gaining competitive advantages since the Taiwan and other governments have established chances that they need local production engagements to access the markets or government procurement contracts.
Global Manufacturing Strategy Implications: The 755 million Taiwan investment is an example of a greater global trend in the pharmaceutical manufacturing strategies on a global scale. Businesses need to consider their manufacturing plants and look into the possibilities of developing regional capacity that captures efficiency in operations and market demands of supply chain resilience and accessibility. The period of global supply chains that are cost-optimized is over and there are more intricate approaches that entail geopolitical risk, supply security, and regulatory compliance in addition to the conventional considerations of cost.
- Eleven Major Pharmaceutical Companies Face Congressional Pricing Transparency Demands
Democrat senators, headed by Ron Wyden, formally requested that AbbVie, Amgen, Bristol Myers Squibb, Genentech, Gilead Sciences, GSK, Johnson and Johnson, Merck, Novartis, Sanofi, and other industry powerhouses should present tangible proof that drug pricing deals of the Trump era do in fact lower prices to Medicaid schemes. This congressional investigation is specifically related to Most Favored Nation agreements of pricing, and therefore, exact disclosure of covered drugs, accurate pricing terms and quantifiable amount of savings that the government healthcare programs are projected to gain.
Escalation in Regulatory Risk: The eleven companies combined have a hundreds of billions of annual revenue in the United States, and any alteration in its pricing policy could be substantial to the financial results. Businesses are currently facing the difficult situation of showing pricing concessions without setting precedents that might bring in more regulations or compromise the business plans in the private insurance markets. The lack of convincing evidence of Medicaid savings would prompt fresh legislative initiatives of compulsory prices or greater power by the government in negotiating.
Compliance and Transparency Requirements: This question is an indicator of the ongoing political attention to the transparency of pharmaceutical prices that may not be limited only to the first group of intended companies. To illustrate value propositions and affordability programs, organizations are supposed to prepare detailed documentation of pricing policies, rebate programs, and actual net prices in various payer channels. It has imposed on manufacturers the burden of demonstrating that international pricing benchmarks provide real benefits to the taxpayers of the United States and government programmes to provide healthcare.
- Pfizer Enters China's Massive Obesity Market with GLP-1 Approval Worth Billions
This allows Pfizer to compete in a market with a potentially tens of billions of dollars each year and was granted regulatory approval in China with its GLP-1-based treatment of obesity, with Pfizer as its partner in China, Sciwind Biosciences. The Chinese obesity drug market is a huge business opportunity with the level of obesity among the populations running into the hundreds of millions and demand that one day may be comparable to the markets of the west in the global GLP-1 segment projected to reach more than 50 billion dollars in sales annually.
Competitive Market Entry: The acceptance allows Pfizer to disrupt the duopoly of duopoly dominance in the largest market in the world between Novo Nordisk and Eli Lilly. The collaboration with Sciwind indicates a more vital approach that multinational firms in China must adopt as it operates in a challenging regulatory structure in which local biotech alliances can hasten the approval processes and offer much-needed market intelligence and medical system connections.
Global Market Implications: The positive results on entering China will provide justification of the GLP-1 mechanism in treating obesity among the Asian populations as they have different metabolism and other clinical considerations that present special market needs. The acceptance has an effect of consolidating regional regulatory rulings in the Asia-Pacific markets and sets competitive precedents on pricing and market access strategy. The next-generation obesity treatments being developed by companies should now think of how to launch at the same time in the markets of the United States, European, and Asian markets to ensure the competitors are not able to gain their regional markets.
- Eli Lilly Launches Direct Employer Platform to Bypass Traditional Insurance Barriers
Eli Lilly developed a direct-to-employer system that allows employers to directly contract with a company to offer their employees access to obesity drugs that bypass the traditional insurance reimbursement plans that have left many patients unable to afford the drugs, which cost more than a thousand dollars a month. The platform combines prescription management, care treatment, and virtual support services into a unified solution that manages cost by establishing direct relationship with manufacturers and makes implementation easier on the part of employers.
Commercial Model Innovation: This model will solve the underlying market access issues in obesity therapeutics, where insurance cover constraints have become limiting its adoption despite positive clinical evidence and patient demand. Contracting directly with employers also puts Eli Lilly in a position to capture patients who would otherwise not be treated because of insurance barriers, and possibly provide them better pricing than the traditional pharmacy benefit manager channels.
Evolution of the market access: The direct-to-employer model may transform pharmaceutical commercialization of high-cost chronic disease therapies across various therapeutic classes. Provided it is successful, this model can be extended to other specialty drugs with insurance coverage restrictions limiting market penetration despite clinically strong propositions. The topic of obesity treatment is increasingly being discussed by employers as a strategic workforce investment with quantifiable costs and benefits in lower healthcare expenditure and enhanced productivity, as opposed to merely increased healthcare expenditure.
Competitive Response in the Industry: Competitors in the market with the manufacture of other obesity drugs are likely to come up with their version of the direct access programs and this may divide the market into various proprietary distribution channels. The pharmaceutical manufacturers are taking a new form that healthcare providers, pharmacy benefit managers and traditional insurers will need to adapt to a new environment which is characterized by pharmaceutical manufacturers gaining influence over patient access via direct contracting relationships which do not necessarily go through conventional reimbursement mechanisms.
- FDA Resumes Advisory Committee Reviews Starting with AstraZeneca's $2 Billion+ Breast Cancer Market
FDA announced intentions to restart advisory committee meetings with the assessment of oral SERD drug Truqap by AstraZeneca in the treatment of breast cancer, which would be the first external expert consultations in a long time. This first major review choice of an oncological treatment indicates the further regulatory focus on cancer therapies and the growing sophistication of targeted therapies which necessitate special scientific testing.
Regulatory Process Normalization: Advisory committees offer independent scientific review of complex treatments by conducting open dialogues of scientific evidence on the advantages, risks, and suitability of patients. The resume also indicates harder scrutiny by the regulatory bodies and possibly longer approval process as FDA goes back to the regular review procedures with outside specialists consultation on complex targeted therapies.
Implication on the markets: The oral SERD of AstraZeneca is an innovation in the treatment of hormone receptor-involved breast cancer, and it is more convenient than injectable stable treatment options but may show superior efficiency due to higher adherence rates. Its approval would confer considerable strength on AstraZeneca in the multi-billion dollar breast cancer therapeutics market and it will enable oncologists to have valuable new treatment opportunities of patients with endocrine therapy resistance.
Adjustments to the Development Strategy: Firms whose oncology programs are about to undergo regulatory filing must consider adding more time to possible advisory review#### and make sure clinical data packages address probable inquiries regarding patient selection, endpoint validity and long term safety surveillance. This additional scrutiny is an expression of the complexity of the contemporary targeted therapies as well as of the regulatory emphasis on assuring that approved cancer drugs provide meaningful clinical benefits that justify their high costs.
Strategic Market Outlook: $4 Billion in Dealings Are an Indication of Change Industrywide.
All these events of March 6 amount to more than 4 billion dollars in strategic dealings and government investment which reflect underlying changes in the priorities and external forces that define the decision-making of corporations in the pharmaceutical industry. The deliberate dispositioning of the commodity business by Lonza and Sanofi shows how top firms are focusing resources on highly differentiated higher margin segments in which sustainable competitive advantages may be sustained despite an increasing pressure on prices.
Rationalization Acceleration of Portfolio: It appears that companies are ready to compromise income magnitude so that they can attain quality margins and strategic concentration in therapeutic segments that provide defensible competitive edge. This trend is on the verge of increasing as development expenses keep on increasing and pressure on pricing increases in various markets. Companies ought to review their portfolios so as to be able to call out non-core assets that can be sold to finance investment in innovation and special capabilities.
Government Investment in Supply Chain Security: Taiwan has made a significant investment in pharmaceutical supply chain resilience and the amount of 755 million is an indication of government awareness globally that pharma supply chain resilience ought to be heavily invested in by the government. The presence of similar programs in the Asian Pacific, European, and North American market will provide partnership opportunities to companies who are interested in establishing regional manufacturing capacity and potentially act as a barrier to market access to those who are simply pursuing centralized production policies.
Increase in Regulatory and Political Pressure: Review of Pharmaceutical Pricing agreements by congress suggests that a consistent political attention on affordability, that cuts across party lines. Firms would have to establish advanced strategies on how to display value and control the perception of the public such as open reporting of net prices, patient assistance initiatives, and investment in innovations. The capacity to estimate the actual savings of the government programs in the real world will become a vital condition of the positive of the government regulation.
Market Access Innovation Requirements: The achievements of the direct-to-employer obesity drug platforms and China market entry strategies prove that the conventional commercialization methods are not enough to embrace the opportunities in high-growth therapeutic categories. To handle the problem of affordability companies have to be creative in the product development, market access, patient engagement, and demonstration of value.
Such changes are indicative of an industry that is in the process of a calculated change into increased specialization, regional diversification, and new business models and that is confronted with complex regulatory, political, and access to markets issues that demand complex strategic responses than what is otherwise the pharmaceutical business model practices.



