Weekly Pharmaceutical Intelligence Brief: Strategic Developments Reshaping Industry Dynamics
Author:
Intellectual Market Insights Research
Published Date:
24 Mar 2026
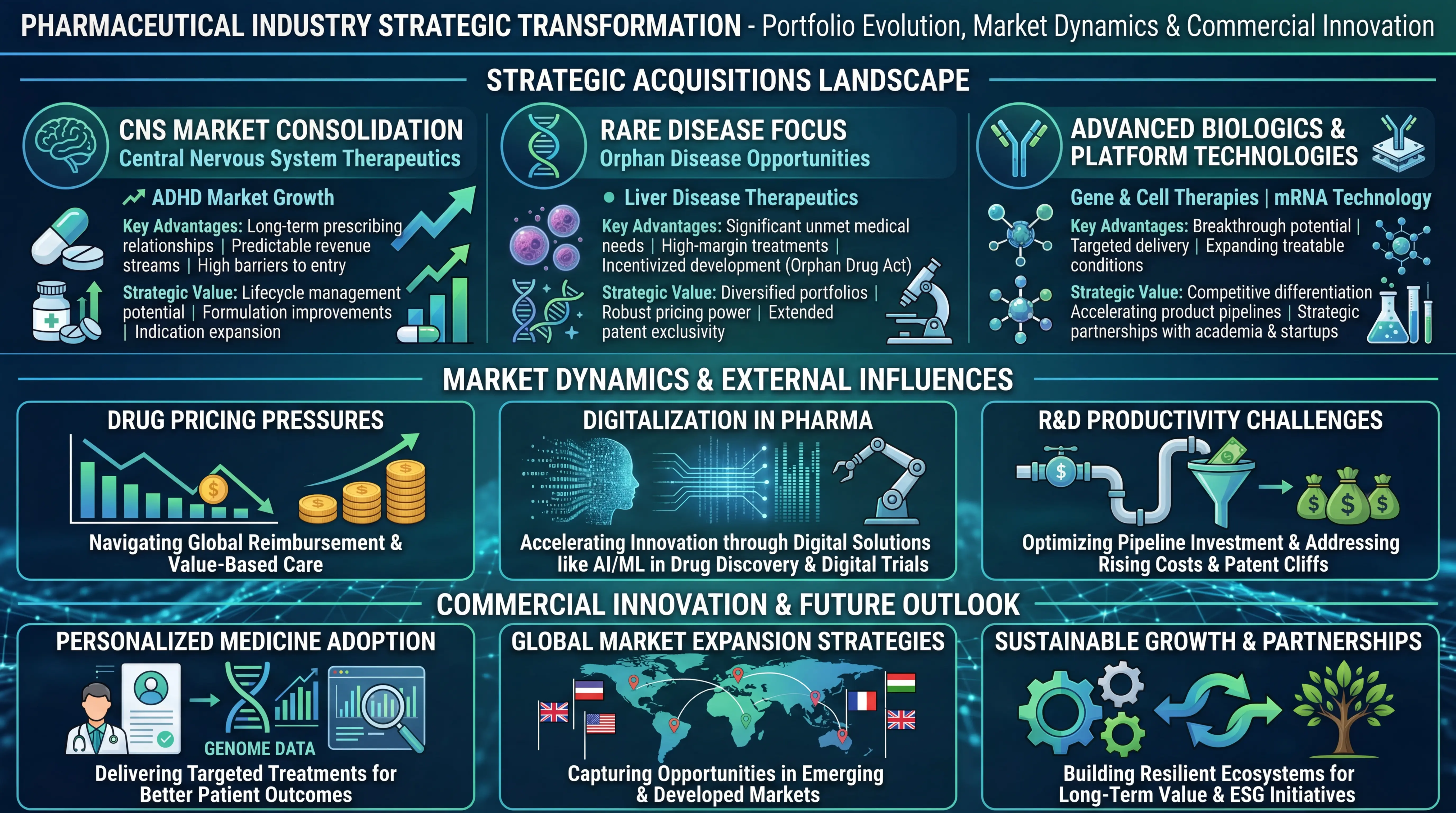
Strategic Acquisitions Drive Specialty Portfolio Expansion
Central Nervous System Market Consolidation
There has been tremendous consolidation behavior in the CNS therapeutic space, with a number of companies engaging in strategic acquisitions to enhance their presence in high-value neurological and psychiatric markets. The appealing basics of CNS therapeutics such as high patient population, high reimbursement coverage, and the possibility of lifecycle management through formulation and indication enhancements are reflected in these moves.
CNS assets have numerous strategic strengths with regard to the purchase of companies. The therapeutic field is characterized by the established trends in prescribing, and the healthcare professionals usually have long-term relationships with the patients who need long-term medication. This provides predictable sources of revenue as opposed to acute care markets. Also, CNS drugs are difficult to develop, which poses natural barriers to entry to existing products against a quick-mover generic development.
The ADHD market in particular is a valuable acquisition target because of its long term growth trend. The rising levels of diagnosis among all ages, the rise in the level of acceptance of the pharmacological interventions, as well as the creation of better formulations have increased the market to be addressed. The businesses that purchase ADHD-related assets have an opportunity to use established commercial infrastructure and diversify their revenues and reliance on single-product franchises.
Rare Disease and Specialty Care Focus
The tendency to purchase rare diseases demonstrates the essential changes in pharmaceutical policy and the market trends. Companies are also becoming more aware of the fact that orphan diseases are attractive investment opportunities even with smaller populations of patients. Such therapeutic segments normally have a high pricing power, longer market exclusivity, and preferable regulatory routes, which may fasten time to market.
In the case of specialty pharmaceutical firms, the rare disease assets fulfill various strategic functions. They offer high-margin income streams that are not prone to pricing pressure and generic rivalry. The nature of these treatments being special also provides a chance to establish further relations with key opinion leaders and academic medical centers which may form platforms to develop pipelines in the future.
Examples of such dynamics are the liver disease therapeutic space. Liver disorders with rare conditions are commonly poorly treated, and there is a very high unmet medical demand. Any therapy that demonstrates meaningful clinical benefit is potentially able to fetch a substantial market value in addition to regulatory incentives such as orphan drug designation which grants regulatory benefits such as expedited review, clinical development tax credit and market exclusivity.
Innovation Strategy Recalibration Toward Near-Term Value
The pharmaceutical sector is proving to be more disciplined in managing the research and development portfolio and the firms are making strategic decisions to eliminate very speculative programs and focusing on more commercially viable opportunities. This pattern depicts the long-standing dilemma of prioritizing the potential of breakthrough innovations and the short-term productivity of the pipeline and the expectations of investors.
The most recent terminations of partnerships in the field of frontier science underscore the nature of risks involved in highly experimental methods of therapy. Although such technologies as advanced biologics platforms and new drug delivery systems have the potential to be transformative, they also pose high levels of technical risk and long development cycles, as well as unpredictable regulatory routes. The move to abandon such alliances may be symbolic of portfolio optimization in which firms reallocate efforts towards programs that have more evident commercialization options.
Such a recalibration is not an indicator that breakthrough science has been abandoned, but a more stringent attitude towards resource allocation. With such stage-gate criteria, companies are demanding more convincing proof-of-concept data during the early-stage programs before they devote considerable resources to it. This will enable the overall portfolio risk to be managed and will still allow the ability to take truly transformative opportunities, should technical and commercial viability be established.
The strategic implications are not limited to individual programs, but to R&D strategy in general. Companies that manage to strike the right balance between innovation ambition and commercial pragmatism will be in a better position to provide some form of pipeline movement without losing investor confidence. Such a rigorous practice of innovation also allows biotechnology enterprises to repurchase rights to potentially profitable technologies and even search new partners with varying risk-taking or strategic interests.
Obesity Market Evolution and Competitive Dynamics
Precision Medicine Positioning in Weight Management
The obesity treatment environment has remained dynamic and firms are competing to find unique ways of gaining value in this growing market. Although general purpose GLP-1 receptor agonists prevail in the current obesity treatment arsenal, the presence of more specialized treatments based on genetically determined patient groups is establishing justifiable market niches.
Competitive advantages of precision medicine in obesity have a number of strategies. With the emphasis on patients who have genetic defects in weight regulation pathways, companies can make their products stand out among mass-market offerings and will have a clear rationale when charging high prices based on the medical necessity. The strategy will also lessen direct competition with blockbuster obesity medicines because the alternative treatment of the previously incurable will be done.
The growth of precision medicine obesity therapies is indicative of same trends of individualized therapeutic interventions. Genetic diagnostics can be a component of business models used by companies, which will establish good rapport with specialized medical centers and develop a single care pathway integrating diagnostic testing and targeted therapy. This approach offers greater predictable price strength as compared to commoditized weight-loss markets and creates a scientific leadership in disease pathogenesis.
In the case of companies that are going to adopt this strategy, expanding the pools of eligible patients by introducing further clinical data and regulatory filings prolongs the market that can be addressed, preserving the benefits of orphan drugs. These treatments are also attractive partnership or acquisition targets to larger pharmaceutical companies since their specialized nature would enable them add to their portfolio of rare diseases.
Patent Expiration and Market Access Transformation
The threat of losing patenting rights of key obesity treatments serves as a turning point to the international weight management therapy. With the expiry of patent protection in major markets, generic and biosimilar products makers are set to introduce alternative products that will essentially change the forces in the market and how patients access the products.
In the case of the originator companies, this shift will require strategic repositioning by lifecycle management programs. Considering the introduction of next-generation combinations, better formulations and diversification to other therapeutic indications can contribute to stay at the competitive position in the case of generic competition development. Firms are also investing in real world evidence production to prove better clinical outcomes as well as justify high prices of branded products.
The generic competition will radically increase the exposure of obesity treatment around the world, especially in price-elastic markets where the existing prices have prevented uptake. This democratization of access might greatly expand the treated patient base since healthcare systems and patients will be able to have access to cheaper options. The generic manufacturers with the knowledge base of producing complex peptides and existing international network of distributors are well placed to increase the scale of production and compete favorably on price without sacrificing on quality.
This is a development that also portends larger effects on the development of obesity drugs. With the first-generation products being subjected to a generic rivalry, the accent is more on differentiation, in terms of clinical superiority, better safety profiles, or more convenient administration instead of first-mover advantage per se. The companies that invest into the next-generation treating obesity must show obvious clinical benefits to ensure higher prices in the market which is becoming more and more competitive.
Commercial Strategy Evolution in the Digital Era
Omnichannel Engagement Transformation
The marketing of pharmaceuticals is radically changing with companies implementing complex omnichannel techniques that are a combination of connected TV, over-the-top streaming, and traditional media platforms. This change in the trends of media consumption by patients and healthcare providers serves to indicate that the pharmaceutical marketer must create more advanced, data-driven engagement strategies.
The pivot to omnichannel marketing also leaves pharmaceutical firms with more targeted, customized messaging opportunities than it had previously been possible to provide through numerous touchpoints during the patient and prescriber life cycle. The opportunities provided by connected TV and OTT platforms cannot be matched by traditional broadcast advertising, such as the ability to target the audience accurately, measure performance in real time, and modify the campaign dynamically, based on the response data.
In the case of healthcare provider engagement, omnichannel strategy can allow pharmaceutical companies to organize the digital interactions, face-to-face interactions, virtual interactions, and delivery of educational materials in a way that does not jeopardize the time limit of physicians but rather presents valuable clinical information. This combined method enhances the strength of message consistency, a higher level of engagement, and a more profound understanding of the communication channels and content types to promote the prescribing behavior to the greatest possible extent.
The omnichannel capabilities investment also prepares the pharmaceutical companies to survive further changes in the healthcare delivery models, such as the growing use of telemedicine and direct-to-consumer healthcare services. With the growing influence of patients in the treatment process and the digitalization of healthcare, pharmaceutical marketers who can master omnichannel engagement will achieve great competitive advantages in raising brand awareness, informing stakeholders, and, finally, lead to product adoption.
Return on Investment and Competitive Differentiation
The omnichannel marketing strategies are advanced with better ROI on pharmaceutical commercial expenditure with better targeting and measuring capabilities. With the help of data analytics to target and access particular patient demographic via streaming services and digital platforms, companies will be able to increase patient demand more efficiently and minimize wasted impressions and increase congruency with the real prescribing behavior.
The competitive differentiation potential of better commercial execution is not limited to a single product launch, but it goes over a period of life cycle management. The companies that are able to master the element of omnichannel engagement are able to introduce the new products more efficiently, retain engagement during the commercial life of the products, and compete not only in terms of the clinical profile and the price but also in terms of the quality of communication and the level of support.
Strategic Implications for Industry Evolution
All these developments demonstrate that the pharmaceutical industry has undergone a strategic transition, which is marked by a number of major themes that will determine competitive advantage over the next few years.
Strict Innovation and Portfolio Management.
Firms are proving to be more stringent in making decisions in research and development by balancing the potential of breakthrough innovations with its short-term commercial feasibility. This strict business health in managing portfolios supports in order to streamline the utilization of resources and still be in a position to seize transformational opportunities in the event that technical and commercial viability can be proven.
Rare Disease Specialty and Focus.
These promising principles of these markets (e.g., the potential to charge a high price, lengthy exclusivity, and the positive regulatory track record) are reflected by the strategic focus on specialty therapeutics and on orphan diseases. Investing in specialty care portfolios is a growth strategy that companies have successfully executed by executing specific acquisition and licensing deals to position themselves at a high rate of growth.
Growth by the strength of the market.
With large therapeutic segments coming of age, and generic pressures, the industry is modifying to volume-based expansionity, in which mass access of patients is being as relevant as value-added pricing. The companies that find the right path in this transition and make next-generation products will remain the leaders in the changing therapeutic markets.
Commercial Excellence as Core Competency.
High-level omnichannel marketing is profiling competitive success, influencing the extent to which companies are able to transform research and development accomplishment into actual use and extended income increment. Commercial excellence is an element of creating sustainable competitive advantages in product portfolios and therapeutic areas.
Combined, these strategic changes represent an industry that is shifting towards focal innovation, specialty market richness, expanded patient reach and high order commercialization. Those companies that manage to incorporate all these factors and ensure high clinical development rates will be in the best position to be sustained in the ever more complicated and competitive pharmaceutical world.



