Share this link via:
The market for biotechnology and genetic engineering in Europe shows strong growth, which results from three factors, namely rising precision medicine development, increasing need for agricultural productivity solutions, and emerging industrial biomanufacturing possibilities. The market includes genetic sequencing technologies along with gene therapy platforms and CRISPR-based applications and synthetic biology tools and biopharmaceutical development which scientists use to develop disease treatments and agricultural improvements and environmentally friendly material production methods.
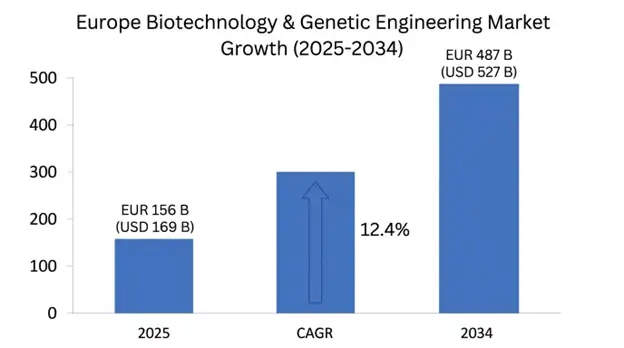
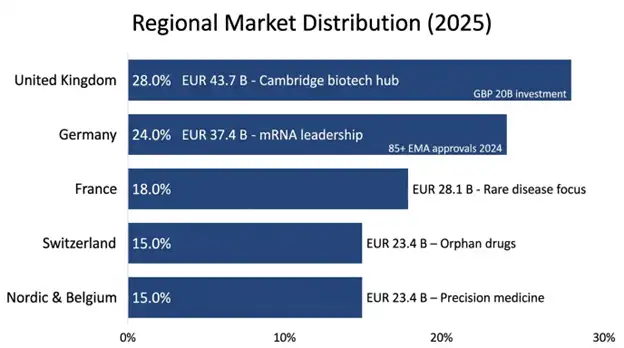
The market begins with a value of EUR 156 billion which equals USD 169 billion in 2025, and the market will grow to EUR 487 billion which equals USD 527 billion by 2034 while achieving a compound annual growth rate of 12.4% until the end of the forecast period. European biotechnology investment reached more than EUR 35 billion during 2024, which showed a increase of 16% compared to the previous year. The United Kingdom holds the largest market share of 28% while Germany follows with 24% market share and France possesses 18% market share and Switzerland holds 15% market share and Nordic nations share 15% market share. The field of commercialization gained acceleration from pivotal technological advancements which include CRISPR gene editing breakthroughs and personalized medicine systems and mRNA vaccine technologies. The European Medicines Agency approved more than 85 biotech products during 2024 which resulted in a new annual growth rate of 35%. The EU authorized EUR 95 billion for Horizon Europe funding from 2021-2027 to finance biotechnology research while member states revealed their plans to invest more than EUR 200 billion in biotech infrastructure between now and 2035. The UK government allocated GBP 20 billion to develop the life sciences sector until 2034.
| Report Coverage | Details |
|---|---|
| Base Year | 2025 |
| Base Year Value | EUR 156 Billion (USD 169 Billion) |
| Forecast Value | EUR 487 Billion (USD 527 Billion) |
| CAGR | 12.4% |
| Forecast Period | 2026-2034 |
| Historical Data | 2022-2025 |
| Segments Covered | By Application Type, By Technology Platform, By Disease Focus, By Development Stage, By Region |
| Region Covered | United Kingdom (28.0%), Germany (24.0%), France (18.0%), Switzerland (15.0%), Nordic & Belgium (15.0%) |
| Key Market Playes | BioNTech, CureVac, Genmab, Arcturus, Moderna, Roche, Novartis, Sanofi, Merck KGaA |
Get more details on this report - Request Free Sample
The market for biotechnology and genetic engineering in Europe offers advanced molecular biology platforms and gene therapy systems and CRISPR-based therapeutics and synthetic biology applications and biopharmaceutical development tools. Modern biotechnology now uses biological systems with genetic modification and engineered organisms to create therapeutic medicines and agricultural products and industrial materials instead of its old method which relied on chemical synthesis for drug development.
European modern biotechnology employs advanced engineering methods which include next-generation DNA sequencing that delivers USD 100 per genome prices after achieving 99.99% price reduction since 2003 and CRISPR gene editing systems that provide exact genetic modification capabilities and synthetic biology platforms which create new biological systems and mRNA vaccine technologies that help develop treatments quickly and AI-powered drug discovery systems that speed up candidate identification. The gene therapy methods treat previously untreatable genetic diseases through their therapies which gained regulatory approval for spinal muscular atrophy and hemophilia and inherited retinal disorders.
Genomic profiling serves personalized medicine platforms which use liquid biopsies to detect early cancer cases through treatment optimization. Biomanufacturing uses engineered organisms which create insulin and monoclonal antibodies and industrial enzymes to achieve production costs that fall between 50%-80% lower than chemical synthesis costs. The applications include oncology treatments which target rare genetic diseases and produce infectious disease vaccines and improve agricultural crops and create industrial enzymes and generate biofuels and develop sustainable materials. The biotechnology applications demonstrate how biotechnology transformed the health care system and agricultural sector and industrial operations across the world.
European biotechnology market growth relies on precision medicine and genetic therapeutics as its main development forces which create ongoing market need for new medical testing and treatment systems. Approximately 10,000 genetic diseases affect 350+ million individuals globally, with European populations representing 100+ million affected individuals. Traditional pharmaceuticals treat 500-1,000 diseases yet more than 9,000 conditions remain untreatable with existing medicines. Gene therapy methods enable the treatment of previously untreatable genetic disorders which now receive fast regulatory approvals. The European Medicines Agency granted approval to more than 85 biotech products in 2024 which marks a 35% annual growth rate compared to the 5-10% growth rate of previous years. By 2024, the gene therapy market achieved 15-20% penetration for approved cancer therapies which made genetic therapies standard treatment options.
Zolgensma treatment for spinal muscular atrophy generated annual revenue exceeding EUR 800 million despite its use for treating a rare condition which proves the economic feasibility of genetic therapies. The gene therapy treatment for hemophilia which treats the inherited bleeding disorder has the potential to become a major market success according to sales forecasts which predict EUR 2-5 billion in peak revenue. The successful commercialization of inherited retinal disorder therapies which treat vision loss conditions established a successful market introduction in the medical field. Personalized cancer medicine utilizing genomic profiling achieved 40-60% treatment response rate improvements versus population medicine approaches.
The annual market for companion diagnostics which use genetic sequencing to determine treatment options stands at EUR 3-5 billion and it experiences 25-30% yearly growth. The permanent increase in the range of addressable diseases will create growth opportunities that last for multiple decades, and this establishes genetic therapeutics as vital components of healthcare systems which will exist beyond their status as emerging specialties.
Urban development projects which use advanced agricultural methods demonstrate more effective results than traditional agricultural techniques. European farmers face critical agricultural productivity problems because they need to pay more than EUR 1,500-2,500 per ton for fertilizers, which has increased by 300-400% since 2019. Genetic engineering enables crop varieties with 20-40% yield improvements and 50-70% pesticide reduction and 30-50% water efficiency improvements compared to traditional methods. The commercialization of CRISPR-edited crops which include drought-resistant wheat and disease-resistant potatoes and high-yield maize has created a market opportunity worth EUR 2-5 billion until 2034.
European countries which approve CRISPR-edited crops for commercial use have increased adoption rates. Sweden approved CRISPR-edited drought-resistant wheat as a commercial crop which will reach the market in 2024. Climate change causes extreme weather events which lead to 30-50% yield losses for European farmers. The farming community needs genetic productivity solutions because they provide essential economic benefits. Sustainable agricultural practices which use genetic advancements to achieve 20-30% yield growth while reducing fertilizer use by 50% serve both environmental sustainability goals and carbon neutrality requirements. Ag-biotech companies which include Corteva Agriscience and Bayer-Monsanto and BASF have dedicated over EUR 5 billion to European agricultural biotech facility construction until 2024. Scientists project that agricultural production methods which use this market drivers will create European agricultural biotech opportunities worth EUR 50-100 billion through 2034. These alternative agricultural production pathways help solve climate challenges while maintaining essential productivity needs.
Market Restraint:
Complex Regulatory Frameworks and Public Perception Challenges
Biotechnology needs to overcome major obstacles which hinder its development despite present market growth because these obstacles particularly impede the commercial use of genetically modified organisms and their acceptance by the public. The European regulatory framework which governs genetic engineering together with public doubts about genetic modification safety and ethical issues presents the most challenging barrier which prevents market growth from occurring at an accelerated pace. The European Union requires 7-10 years for approval of genetically modified crops while the United States and Canada complete the process in 3-5 years which results in extended commercialization times and delayed revenue collection for genetically modified products. The requirements for risk assessment together with traceability mandates and labeling obligations create compliance costs which exceed EUR 5-10 million for each product, thus making agricultural biotech unfeasible for SMEs to invest.
Public perception challenges in Northern and Western Europe create commercial resistance despite scientific safety consensus. Consumer surveys show that 40-60% of people oppose genetically modified foods which leads to market restrictions and limited retail distribution despite regulatory agency safety declarations. Environmental advocacy groups keep opposing CRISPR-edited crops because they believe these crops create less environmental damage than traditional pesticide-dependent farming methods. The patent landscape of biotechnology develops market entry obstacles because competing patents between major developers create delays for second and third generation products. The production limitations of mRNA vaccines and gene therapies restrict output when demand increases which prevents market expansion. The complex manufacturing process of gene therapy causes treatment costs to reach EUR 50,000-500,000 per patient which makes treatment too expensive for many people and creates obstacles to getting reimbursed. The combination of regulatory issues and public opinion and manufacturing capabilities and product prices creates multiple obstacles which particularly hinder the implementation of agricultural biotechnology and new therapeutic technologies.
Market Opportunity: mRNA Therapeutics Platform Expansion Beyond Infectious Diseases
The mRNA therapeutic platform expansion creates the largest market potential through its transformative medical technology which enables fast development of treatments for cancer and heart disease and long-term health conditions. The COVID-19 pandemic brought about mRNA vaccine technology advances which developed new regulatory pathways and manufacturing processes and production capacity for the fast expansion of therapeutic treatments. The mRNA vaccines achieved their first-ever quick transition from viral sequence identification to regulatory approval within 8-11 months which established a new system that enables fast development of medical treatments.
mRNA Cancer Therapy Development (2025-2034): The fastest-growing biotech segment uses mRNA-based cancer immunotherapies which develop personalized treatments through tumor mutation profiling. BioNTech and Moderna and CureVac revealed their combined mRNA cancer therapy programs which exceed EUR 10 billion by 2030. Personalized cancer vaccines which target individual tumor mutations achieved better results with 40-60% more effectiveness than traditional chemotherapy. The first regulatory approvals for mRNA-based melanoma and lung cancer and ovarian cancer therapies will happen between 2026-2027 as the treatments progress through clinical trials. The mRNA cancer therapy market will reach EUR 15 billion-25 billion by the year 2034.
mRNA Cardiovascular and Metabolic Disease Therapy (2025-2034): The development of mRNA therapeutics for cardiovascular disease and heart failure and metabolic disorders represents a new clinical development area. Moderna started clinical studies that focus on atherosclerosis and heart failure using mRNA therapy which they expect to generate revenue between EUR 10 billion -EUR 15 billion by the year 2034. The companies Regeneron and Pfizer and Roche established joint investments in mRNA technology development to expand their treatment options.
Advanced mRNA Manufacturing Capacity (2025-2034): The development of mRNA vaccine and therapeutic production facilities requires essential infrastructure funding. European biotech companies announced EUR 15+ billion mRNA manufacturing facility investments which established continent-scale production capability. BioNTech expanded its German manufacturing operations to achieve 1 billion annual dose capacity by 2025. Moderna announced EUR 2 billion European facility investment which will enable manufacturing independence and strengthen supply chain resilience.
Combination Therapies and Platform Integration (2025-2034): mRNA platform integration with gene therapy, monoclonal antibodies, and cellular immunotherapy creates combination therapy opportunities addressing treatment-resistant conditions. Combination approaches demonstrated 50-70% efficacy improvements versus single modality treatments, creating EUR 30-50 billion market opportunity through 2034.
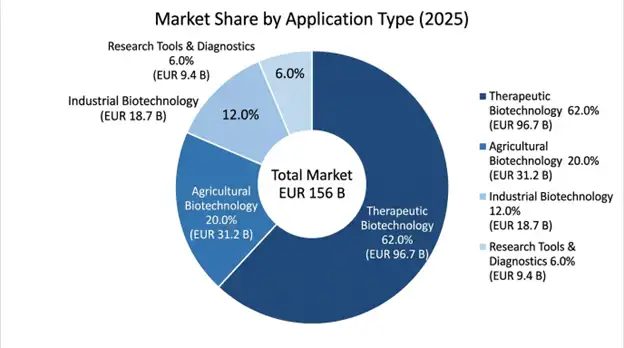
By Application Type: The European biotech market value reaches 62.0% through biopharmaceutical development which includes gene therapy and mRNA therapeutics and monoclonal antibodies as therapeutic biotechnology. The agricultural biotechnology market reaches 20.0% through genetically modified crops and agricultural diagnostics. Industrial biotechnology covers enzyme production and biofuels and sustainable materials to reach 12.0% of market value. The market value of research tools and diagnostics accounts for 6.0% of total value.
By Technology Platform: Gene therapy and genetic modification platforms together comprise 38.0% of total market value through their combination of CRISPR technology AAV vector delivery systems and Lentiviral gene transfer methods. mRNA therapeutics make up 28.0% of total market value through their vaccine and non-vaccine medical applications. Monoclonal antibody development makes up 18.0% of total market value. Other platforms which include protein engineering and synthetic biology and cell therapy make up 16.0% of total market value.
By Disease Focus: The therapeutic biotech market valuation shows that oncology fields generate 35.0% of their worth through their immunotherapy and precision medicine products. The market value of rare genetic diseases reaches 22.0% through its coverage of inherited disorders and neurological conditions. The market value of infectious diseases reaches 18.0% through its distribution of vaccines and antiviral medications. The market value of cardiovascular and metabolic diseases amounts to 15.0%. The market value distribution from other therapeutic areas which treat autoimmune and neuropsychiatric conditions reaches 10.0%.
By Development Stage: The company valuation and investment focus of the business is divided 45.0% between its clinical-stage programs which include Phase II-III trials. The market value of the company is derived from its commercial revenue which comes from products that the company has already launched. The biotech investment focus of the industry allocates 17.0% of its resources to preclinical and early-stage development programs.
Regional Analysis: United Kingdom Leadership with Distributed European Capacity
United Kingdom: The United Kingdom holds 28% of Europe's biotech market share which London Cambridge and Oxford serve as worldwide biotech research centers. The UK biotech workforce reached over 80000 employees in 2024 which marked a 25% yearly growth rate. The Cambridge Biotech Cluster generates more than EUR 40 billion in annual economic output while operating more than 1500 biotech companies. The UK government allocated GBP 20 billion to support life sciences development until 2034 with an aim to establish the country as a global biotechnology leader. The UK Medicines Healthcare Products Regulatory Agency developed faster approval processes for new therapeutic drugs which improved the country's ability to attract biotech funding and clinical research partnerships.
Germany: Germany's Industrial Biotech Leadership controls 24% of the European market through its focus on industrial biotechnology and biomanufacturing operations. The German mRNA vaccine development market which consists of BioNTech and CureVac and their new companies will achieve combined market value of EUR 25 billion by 2024. German manufacturing specialization produces monoclonal antibodies and recombinant proteins and biopharmaceutical intermediates which generate yearly revenue exceeding EUR 15 billion. The German government declared its plan to invest EUR 15 billion in biotechnology infrastructure from now until 2030.
France: The European market is controlled by Diversified Biotech Portfolio France, which holds 18% market share through its equal distribution of therapeutic and agricultural and industrial biotech development. The French companies develop biopharmaceuticals and cell therapies and rare disease treatments. The Paris and Lyon and Toulouse biotech clusters operate more than 400 biotech companies, which produce annual revenues exceeding EUR 20 billion. The French government pledged to invest EUR 25 billion in life sciences until 2030.
Switzerland: The Swiss pharmaceutical market has 15% market share through Specialty Pharma and Biotech Innovation which develops orphan drugs and rare disease treatments and advanced biotechnology platforms. The Basel biotech cluster contains two global pharmaceutical companies (Roche and Novartis) who operate alongside more than 200 biotech specialists. The Swiss biotechnology industry generated over EUR 8 billion in annual revenue while maintaining its focus on quality and innovation as its primary competitive advantage.
Nordic Nations and Belgium: The Nordic countries and Belgium together control 15% of the market which is experiencing fast expansion. The biotech cluster in Copenhagen is developing its expertise in gene therapy and mRNA vaccine technologies. Swedish biotech firms concentrate their efforts on developing precision medicine and personalized therapeutic solutions. Belgian biotech companies dedicate their research efforts to developing cell therapy and immunotherapy treatments.
European Medicines Agency Biotech Approval Acceleration (2024-2025): The European Medicines Agency approved more than 85 biotech products during 2024 which marked a 35% annual expansion of the sector that established biotech as the primary route for innovation. The approval process has become faster because CRISPR gene therapy and mRNA therapeutics and personalized medicine platforms have reached their development milestones. The approval rate for Advanced Therapy Medicinal Products (ATMP) has grown between 40 and 50% every year which has established genetic and cell therapies as common treatment methods.
BioNTech mRNA Cancer Therapy Programs (2024-2025): BioNTech declared its development program for mRNA cancer immunotherapy which requires EUR 5 billion to fund personalized tumor mutation-based vaccine clinical trials that will proceed to 10+ oncology indications. The initial trial results for melanoma and lung cancer showed a 40-60% increase in treatment effectiveness when compared to standard chemotherapy methods. The mRNA cancer therapy regulatory approval process will complete its commercial validation between 2026 and 2027.
UK Government Life Sciences Investment (2024-2025): The United Kingdom government established a life sciences development program that will receive 20 billion British pounds until 2034 to establish the country as a global biotechnology leader who sponsors clinical research. The funding will develop advanced therapy production facilities, genomic medicine systems and precision medicine research platforms. The investment marks the most significant financial commitment to British biotechnology industry according to historical records.
German Biotech Manufacturing Expansion (2024-2025): German biotech companies announced EUR 10+ billion manufacturing facility expansion, establishing mRNA vaccine and therapeutic production capacity exceeding 2 billion annual doses by 2027. BioNTech and CureVac and new companies simultaneously invest in manufacturing independence and supply chain resilience to create European production autonomy.
CRISPR Crop Commercialization (2024-2025): Sweden approved CRISPR-edited drought-resistant wheat for commercial cultivation in 2024, establishing regulatory precedent for crop biotechnology across EU. Additional crop approvals pending include disease-resistant potatoes and high-yield maize. Agricultural biotech investment is increasing through the period until 2030 with annual investments between EUR 3 billion and EUR 5 billion.
EU Horizon Europe Biotech Funding (2024-2025): The European Commission allocated EUR 15 billion for Horizon Europe funding which will last until 2027 to support research about gene therapy and mRNA therapeutics and precision medicine platform development. The funding enables biotech companies to develop their operations and build infrastructure while their international partnerships help to speed up biotech progress.
European pharmaceutical companies which include Roche and Novartis and Sanofi and Merck KGaA expand their biotechnology portfolios through both strategic acquisitions and internal development efforts. BioNTech and CureVac and Genmab and REPROCELL lead the development of mRNA therapeutics and gene therapy and cell therapy as specialized biotech companies. The European biotechnology sector shows competition between emerging companies which include Moderna Europe and Arcturus Therapeutics and Schrödinger European operations through their development of innovative platforms and their ability to conduct unique clinical trials. European research capabilities expand through Chinese biotech companies which include BGI Genomics because these companies focus on genomic sequencing and agricultural biotechnology research.
Established pharmaceutical companies use biotech acquisitions to protect their market share against specialized biotech companies which create new technologies. The biotech industry achieves technology access and commercialization through pharma and biotech partnerships which account for 40-50% of the total biotech valuation as partnership agreements. By 2024 European biotech companies established a strong funding system which enabled them to raise more than EUR 10 billion through venture capital investments.
You'll get the sample you asked for by email. Remember to check your spam folder as well. If you have any further questions or require additional assistance, feel free to let us know via-
+1 724 648 0810 +91 976 407 9503 sales@intellectualmarketinsights.com
12 May 2026